Blog post written by Philipp Boersch-Supan and Cat Lippi from University of Florida.
VectorBiTE scientists from the QDEC Lab Group at the University of Florida have now published a study on seasonal and daily patterns in human biting rates of mosquito taxa capable of malaria transmission in Southern Ecuador. In collaboration with SUNY Upstate Medical University and the Ecuadorian Ministry of Health, the data for this study were collected during a massive effort to eradicate local transmission of malaria in Ecuador’s Southern coast during the 2000’s, resulting in successfully eliminating autochthonous transmission in this region. Although no transmission of malaria has been detected in Ecuador’s southern coast since 2011, mosquito-borne diseases such as malaria, dengue, and other viruses remain a major threat to people’s livelihoods throughout Latin American, which makes it crucial to prevent reestablishment of these diseases where local infection has been eliminated.
Surveillance is key to assessing and maintaining elimination status. Because it is extremely difficult to directly monitor mosquito populations and the pathogens carried by them, vector-borne diseases are often monitored through case numbers of human infections. This adds an additional layer of uncertainty: Infections may be asymptomatic, and access to health care is unevenly distributed, leading to reporting biases. Furthermore, once local elimination is achieved, relying on human case reports means reestablishment can only be detected after transmissions and infections have occurred, leaving limited options for preventative vector control measures. It is therefore important to not only monitor infections themselves, but also the potential for mosquito-borne transmission, or the risk of infectious bites on humans. This risk is often quantified by observing human bite rates or human landing rates. While these do not directly measure infections, they are often cited as a proxy for species presence, density of blood-seeking females, and thus the capacity for disease transmission.
Collecting data on human biting rate in the field poses an ethical dilemma, as field technicians are exposed to the risk of disease transmission. Bite rates in this study were estimated from human landing catch (HLC), where technicians equipped with stockings collected mosquitoes with an aspirator as they landed. These counts were taken hourly both inside and outside of residential buildings across five localities. Because not all mosquito taxa occur at all times and at all study sites, such datasets often exhibit a large proportion of zeros, i.e. observational periods where the observer was not bitten at all by one or more taxa. Elsewhere the observer may sit near a site that is favourable for mosquito breeding and experience many bites in an observational period. Such data are challenging to model with traditional statistical models, because the surplus zeroes, as well as the occasional large counts are not captured by commonly used probability distributions, lead to biased estimates of mean biting rates.
Ecologically the observed quantity (realized bites) arises from a two part process; mosquitoes must be present and able to bite for there to be any bites to count.
We therefore used a two part statistical model to analyze the data, a so-called hurdle model. Hurdle models specify one process to model the probability or odds for zero counts and another process for the positive bite count or rate. The idea is that positive counts occur once a threshold – the hurdle – is crossed. If the hurdle is not cleared, then we have a count of zero.
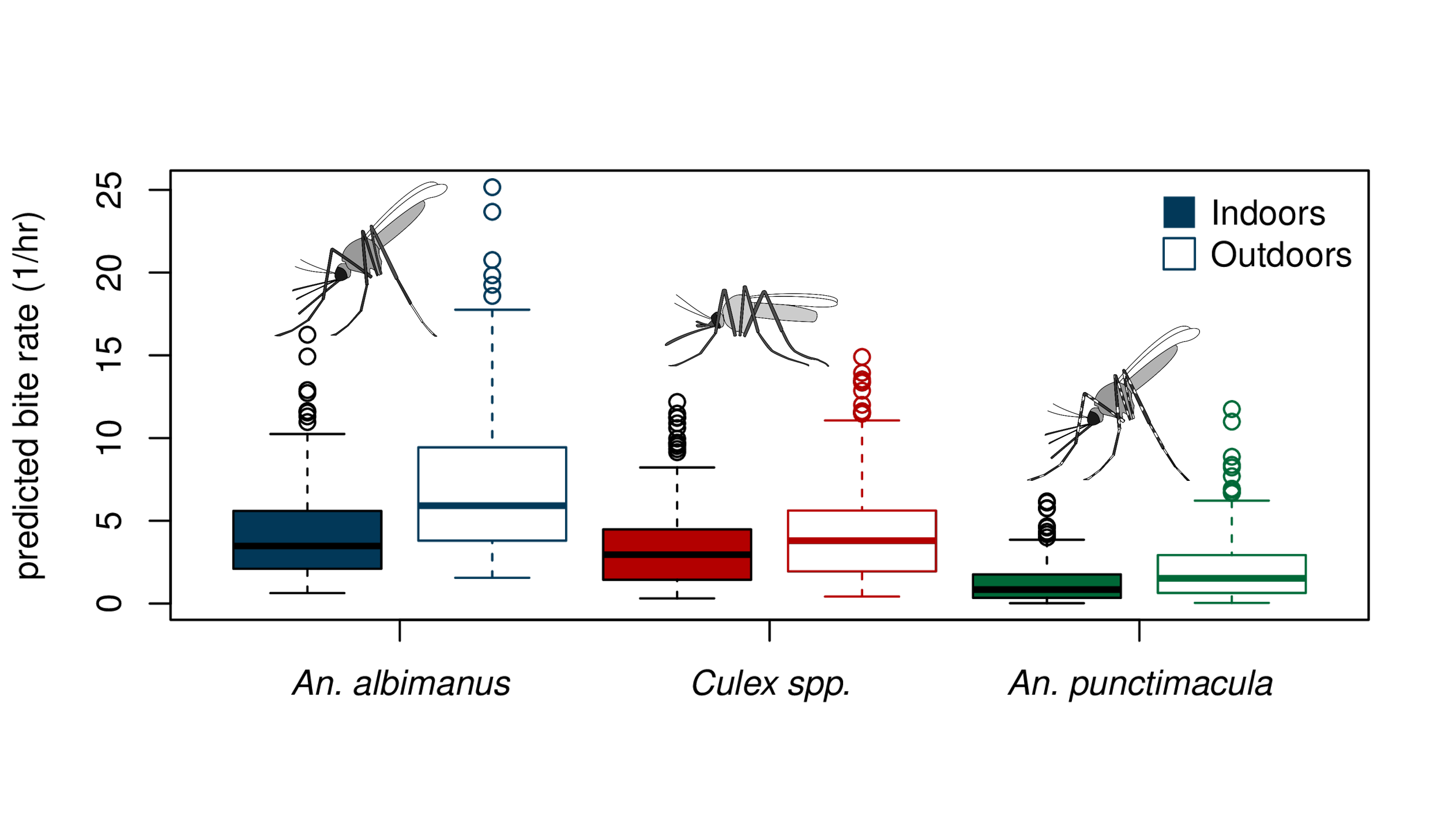
The models allowed us to determine whether biting activity was fundamentally different for different taxa, and to identify temporal and spatial factors (being inside or outside a residential structure) influencing bite rate. We were interested in these potential differences because vector competence (i.e. the ability to transmit malaria) differs among mosquito taxa. We found that biting rates varied significantly among species and time. Anopheles albimanus was the species most commonly observed biting. The occurrence of An. albimanus bites in a given hour was four times as likely as no bites, with an average of 4.7 bites per hour. All taxa exhibited exophagic feeding behavior, meaning that outdoor locations increased both the odds and incidence of bites across taxa.
Being outdoors more than doubled the odds of being bitten by An. albimanus and increased the number of bites per hour by about 50%. Differences between indoor and outdoor locations were less pronounced for the two other taxa, both concerning the odds of receiving any bites and the number of bites received. Months of peak high and low biting activity varied for the three taxa; the highest and lowest respective months for significant biting activity were March and July for An. albimanus, July and August for An. punctimacula, and February and July for Culex spp.
Because vector competence is not the same across all mosquito taxa, our results have important implications for vector control. Aggregating bite rates or other measures of mosquito activity across taxa potentially obscures species-specific peaks of activity. Vector control efforts that are based on aggregated counts may therefore not effectively target the most competent vectors at their peak activity, and by the same token, epidemiological models based on aggregated activity measures may be less accurate in predicting disease outbreaks. Furthermore, the differences observed in biting activity across taxa indicate a need for accurate species descriptions in regions with multiple vectors, as taxonomic confusion between closely related mosquitoes may obscure patterns in transmission risk. While the collection of species-specific bite rate data requires more observer effort, the information provided by the surveillance and analysis methods in our study can be incorporated to allow more targeted, household-level vector control programs, thus providing a powerful tool in controlling and preventing the reemergence of pathogens.